An atom is the smallest unit of matter that retains all of the chemical properties of an element. Atoms combine to form molecules, which then interact to form solids, gases, or liquids.
The atomic structure of an element refers to the constitution of its nucleus and the arrangement of the electrons around it. Primarily, the atomic structure of matter is made up of protons, electrons and neutrons.
Nucleus in the Atom
The center of an atom is called the nucleus and is made up of both protons and neutrons. This part of the atom is able to determine a wide range of properties, such as the atomic number and atomic mass.
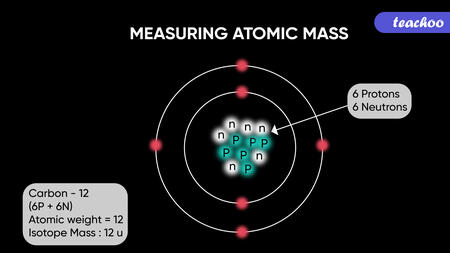
Atomic Mass
The mass of protons and neutrons is roughly equal, weighing 1.67 × 10-24 grams. One Dalton or one atomic mass unit (amu) is how scientists define this quantity of mass. Protons and neutrons have the same mass, but protons are positively charged and neutrons are not. As a result, an atom's mass is greatly influenced by its neutron count, but not its charge.
Electrons weigh only 9.11 × 10-28 grams, or around 1/1800 of an atomic mass unit, which is significantly less than protons. As a result, their contribution to the total atomic mass of an element is the smallest.Atomic mass is the sum of the masses of protons, neutrons and electrons.Atomic mass = Mass of protons + Mass of neutrons + Mass of electrons
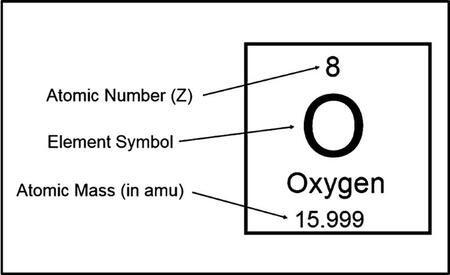
Atomic Number
An element's neutral atoms have the same amount of protons and electrons. The atomic number of an element is determined from its number of protons, which also serves to distinguish it from other elements. For example, carbon’s atomic number is 6 because it has 6 protons.Moreover, the number of neutrons can differ to produce isotopes, which are atoms of the same element that have different numbers of neutrons. The number of electrons can also be different in atoms of the same element, thus producing ions. For example, iron, Fe, can exist in its neutral state, or in the +2 and +3 ionic states.An element's neutral atoms have the same amount of protons and electrons. The atomic number of an element is determined from its number of protons, which also serves to distinguish it from other elements. For example, carbon’s atomic number is 6 because it has 6 protons. Moreover, the number of neutrons can differ to produce isotopes, which are atoms of the same element that have different numbers of neutrons. The number of electrons can also be different in atoms of the same element, thus producing ions. For example, iron, Fe, can exist in its neutral state, or in the +2 and +3 ionic states.
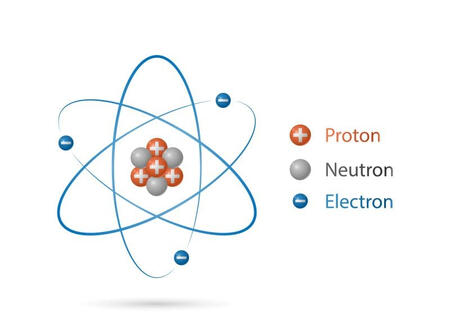
Protons, neutrons, and electrons are the three fundamental subatomic particles that make up an atom. The protons and neutrons are located in the center of the atom, while the electrons are quite far from the center.The number of protons in the nucleus of an atom determines its atomic number and its identity as a specific element. For instance, the nucleus of every hydrogen atom has one proton; on the other hand, the nucleus of every carbon atom contains six protons. The atomic mass of an atom is typically equal to the sum of its protons and neutrons, however the amount of neutrons in the nucleus might vary. Atoms are constantly interacting with each other and can combine to form molecules, which are the building blocks of most of the matter that we encounter in our everyday lives.
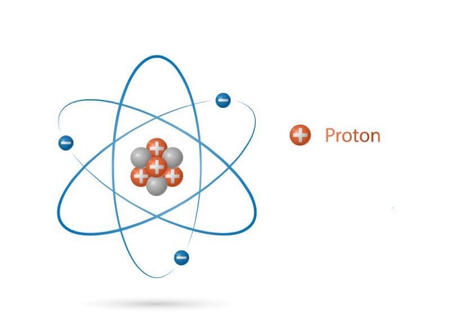
PROTON
They are positively charged subatomic particles, found in the nucleus of all atoms. A proton has a charge of +1, and has a defined mass of 1.007277 amu. The symbol for the proton is either p or p+. The number of protons in an element's nucleus is its atomic number. In addition, free protons exist, the nucleus of a hydrogen atom or the H+ ion is an example of a proton.
NEUTRON
They are neutrally charged subatomic particles, found in the nucleus of all atoms except hydrogen with a symbol n. A neutron's mass is more than a proton's which is 1.008665 amu. Neutrons are very dense and have neither a positive nor a negative electrical charge. The main function of neutrons is to contribute to the binding energy or nuclear glue that holds the nucleus itself together. Furthermore, neutrons are important because they determine the isotope of an element.